General Information
Strain Name | Hu-HSC-NPG |
Origin | Beijing Vitalstar Biotechnology Co., Ltd. |
Background | NPG |
Coat Color | Albino |
Development
Hu-HSC-NPG is a model where irradiated bone marrow-depleted NPG mice are transplanted with human umbilical cord blood and fetal liver hematopoietic stem cells (HSC). After transplantation into NPG mice, the HSCs can engraft in the mouse bone marrow and continuously produce various hematopoietic and immune cells, such as T cells, B cells, NK cells, myeloid cells, etc. Since the immune cells develop "de novo" within the mouse host, they are tolerant to the mouse, and generally do not exhibit Graft-versus-host disease (GvHD) (although some may appear after 30 weeks), allowing for a research window period of up to one year. The specific construction method is as follows:
Severely immunodeficient mice NPG are subjected to sublethal irradiation to destroy the mouse's own bone marrow hematopoietic function, and then transplanted with human umbilical cord blood, bone marrow, or liver-derived hematopoietic stem cells via intravenous injection, bone marrow cavity injection, or injection into the livers of newborn mice, etc. After 12 weeks, the reconstitution of human-derived blood cell lineages is detected in the blood to determine the ratio of human/mouse cells.
Phenotype
1. Survival curve of HuHSC-NPG
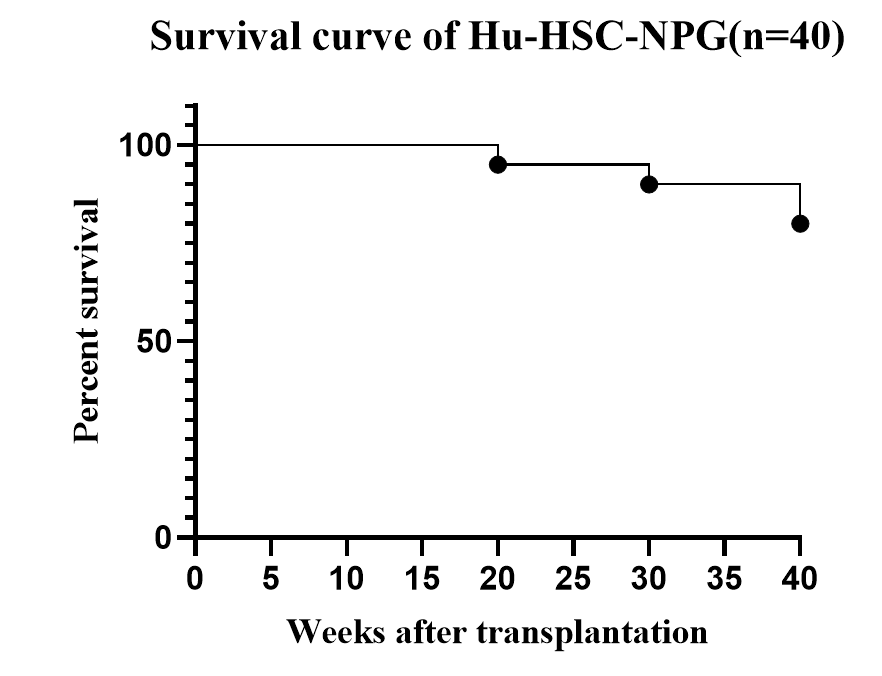
Fig1. Survival curve of HuHSC-NPG
Long-term observation of the survival status of HuHSC-NPG mice, which can exceed 40 weeks, and due to the immune cells developing "de novo" within the mouse host, they are tolerant to the mouse, thus generally not exhibiting graft-versus-host disease (GvHD) (although some may occur after 30 weeks), allowing for a research window period of up to one year.
2. Engraftment of human CD34+ HSCs in NPG to reconstitute human immune system
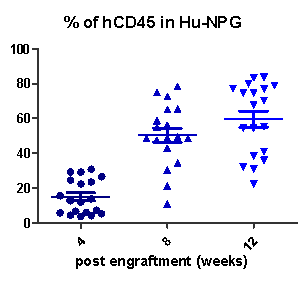
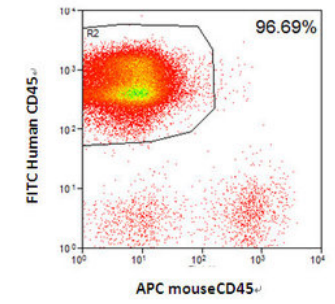
Fig2. Engraftment of human CD34+ HSCs in NPG mice successfully reconstituted human T cells.
Collect peripheral blood from Hu-HSC-NPG mice at different reconstitution periods and analyze by flow cytometry. After transplanting 5×104 umbilical cord blood CD34+ cells into NPG mice, the hCD45+ levels continuously increase with the extension of the reconstitution period (left graph); after transplanting 1×105 umbilical cord blood CD34+ cells, high levels of hCD45+ cells can be detected in the spleen of NPG mice at 16 weeks (right graph).
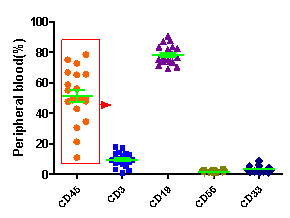
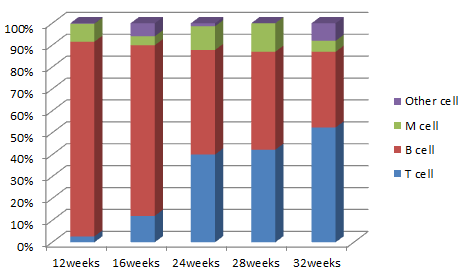
Fig3. Levels of immune cell differentiation at different reconstruction times in HuHSC-NPG mice.
Collect peripheral blood from Hu-HSC-NPG mice at different reconstitution periods and analyze by flow cytometry. After transplanting 5×104 umbilical cord blood CD34+ cells, high hCD45+ leukocyte reconstitution was detected in the peripheral blood of NPG mice at 12 weeks, with the main differentiation into hCD19+ B cells as well as CD3+ T cells and CD33+ myeloid cells (left graph); as the reconstitution period increases, the proportion of T cells becomes higher and higher (right graph).
3. Orthotopic tumor model of HSC-NPG mice
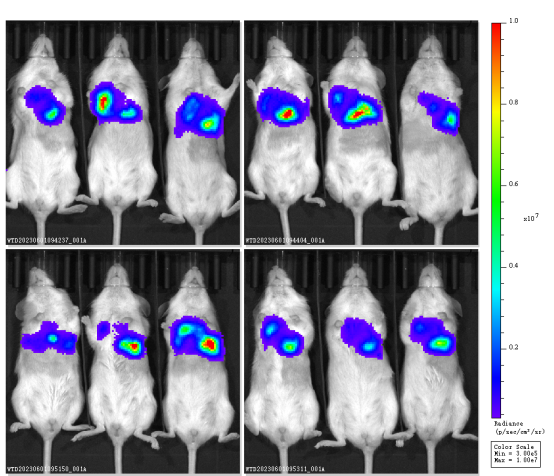
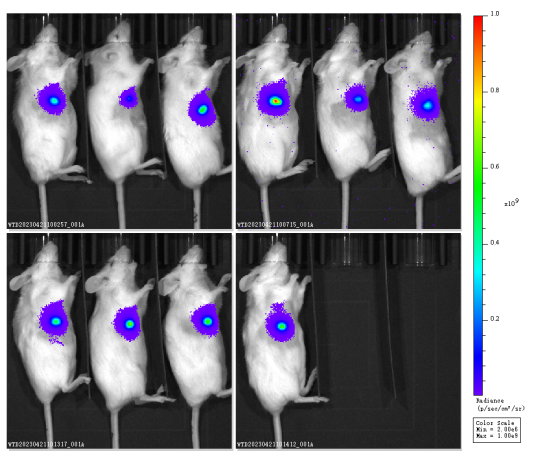
Fig4. Orthotopic tumor model of HuHSC-NPG mice
Hu-HSC-NPG mice transplanted with various tumors and examples of efficacy studies
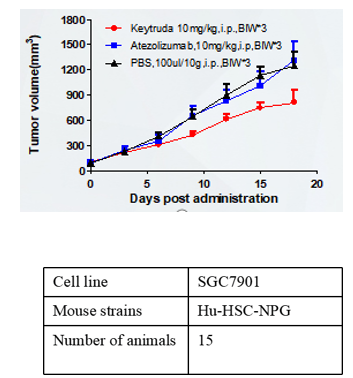
1. Evaluation of different xenotumor transplantation models and the efficacy of antibody
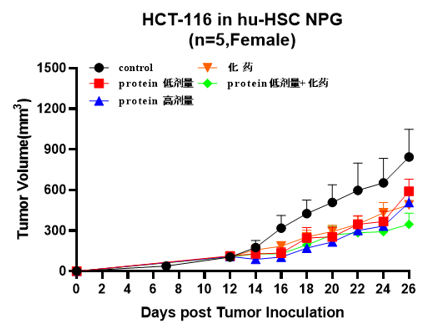
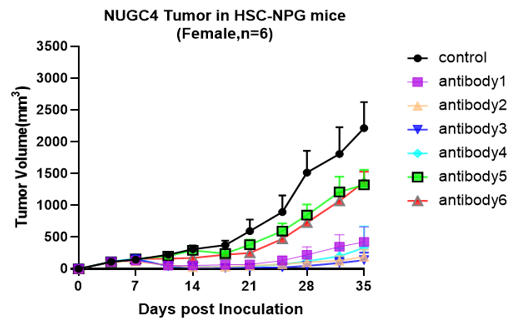
A B
C D
Fig5. Antitumor effects of different drugs
A: Human colon cancer cell line, B: Human gastric cancer cell line, C: Human choriocarcinoma, D: Human glioma
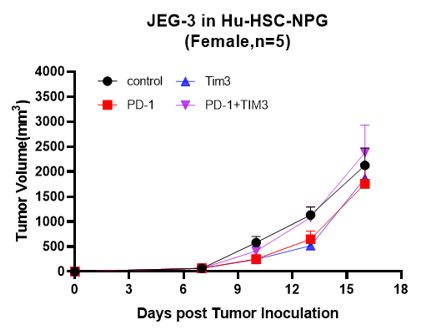
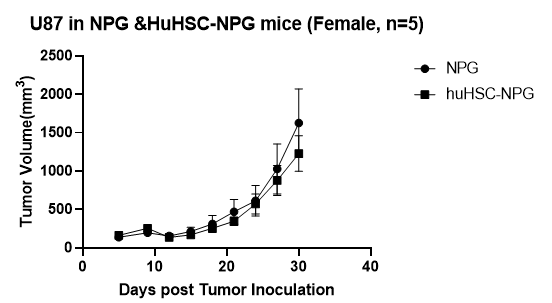
2. Evaluation of different xenotumor transplantation models and the efficacy of PD1/PDL-1 inhibitors
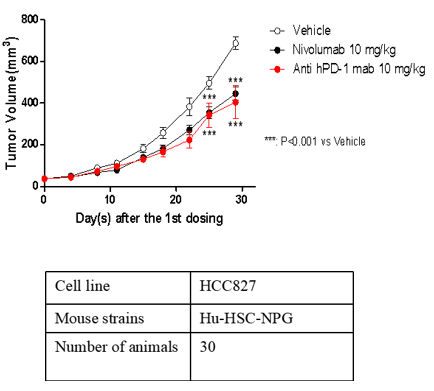
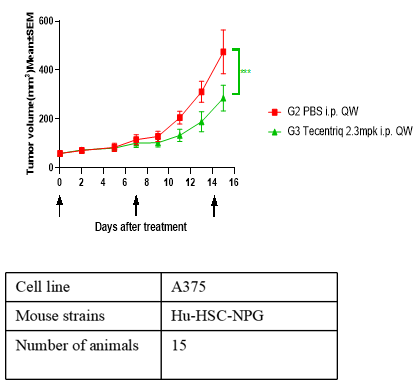
A B C
Fig6. Antitumor effects of different drugs
A: human gastric cancer cell line, B: human non-small cell lung cancer cell line, C: human malignant melanocytic pheochromocytoma.
3. Constructing human gastric cancer model and evaluating therapeutic effect of gastric cancer mesenchymal stem cells combined with PD-1 antibody[1]
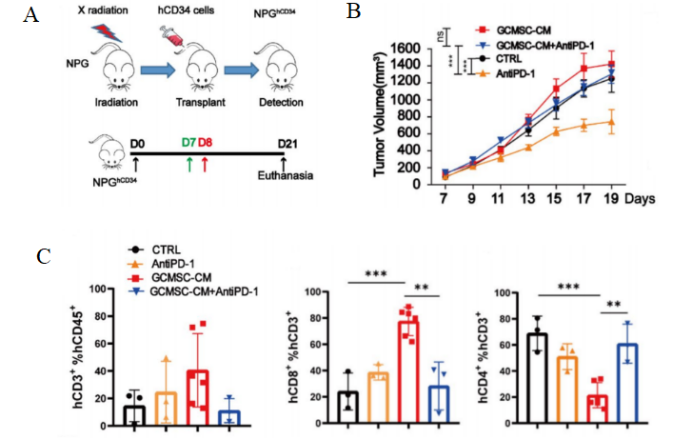
Fig7. Tumor and tumor tissue immune cell levels after HuHSC-NPG inoculation of gastric cancer tumors.
4-8 week-old NPG mice were irradiated with X-rays (1.2 Gy) and within 24 hours intravenously injected with 5×104 human umbilical cord blood CD34+ cells. After 12 weeks of reconstitution, patient-derived gastric cancer cell lines were inoculated, and treated with GCMSC-CM and PD-1 antibody alone or in combination to observe tumor changes, and to detect the levels of CD45+CD3+, CD3+CD8+, and CD3+CD4+ in tumor tissues from different treatment groups. The results showed that after 21 days of treatment, PD-1 antibody therapy significantly inhibited gastric tumor growth, while the combination of GCMSC-CM with PD-1 antibody significantly increased tumor volume compared to PD-1 antibody alone, indicating that GCMSC-CM negated the inhibitory effect of PD-1 antibody on gastric tumor growth.
4. Constructing human lymphoma model and evaluating the efficacy of small molecule inhibitors[2]
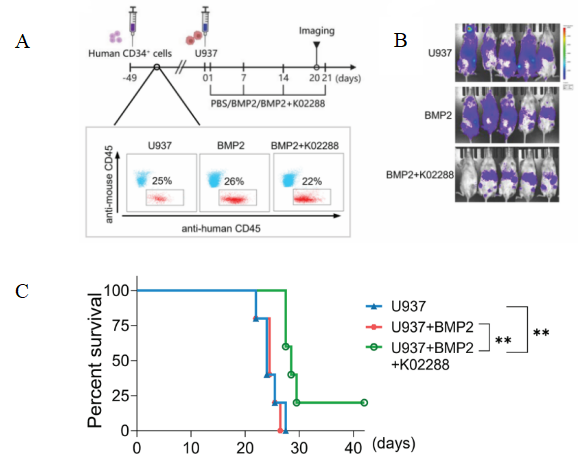
Fig 8. Schematic diagram of HuHSC-NPG inoculated U937 cell construction and anti-tumor effect
Female NPG mice (n=5 per group) at 5-6 weeks of age were irradiated (1 Gy) with 5×105 human CD34+ cells injected into the tail vein after marrow clearing, and the peripheral blood was assayed for antibodies against anti-mouse CD45 and anti-human CD45 after 6-7 weeks. The proportion of human CD45+ cells in the peripheral blood mononuclear cells of mice was > 20%, confirming the success of human leukocyte transplantation. U937 with BMP2 or BMP2 with k02288 were injected as shown. aiming at hindering the tumor growth effect of Reg-Vδ2 T cells by inhibiting the BMP2 signaling pathway through small molecule inhibitors. The results showed that k02288 combined with BMP2 treatment then significantly inhibited the growth and significantly prolonged the survival of U937 cells.
5. Toxicologic evaluation of dual-antagonist drugs[3]
A B

C
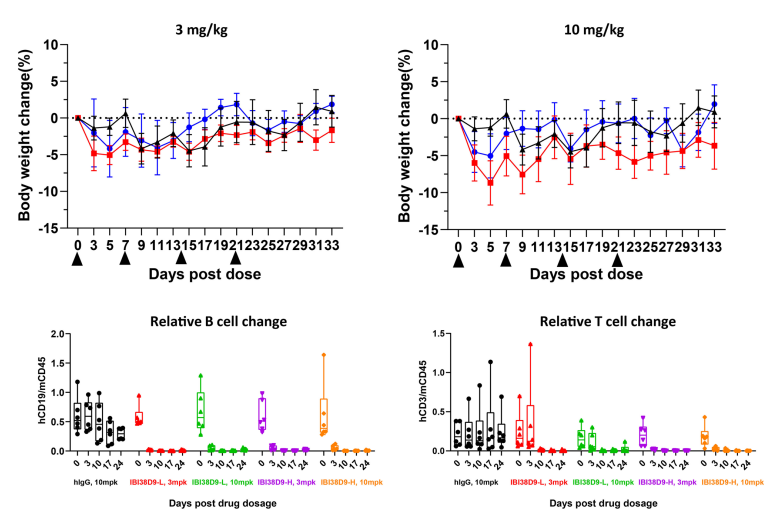
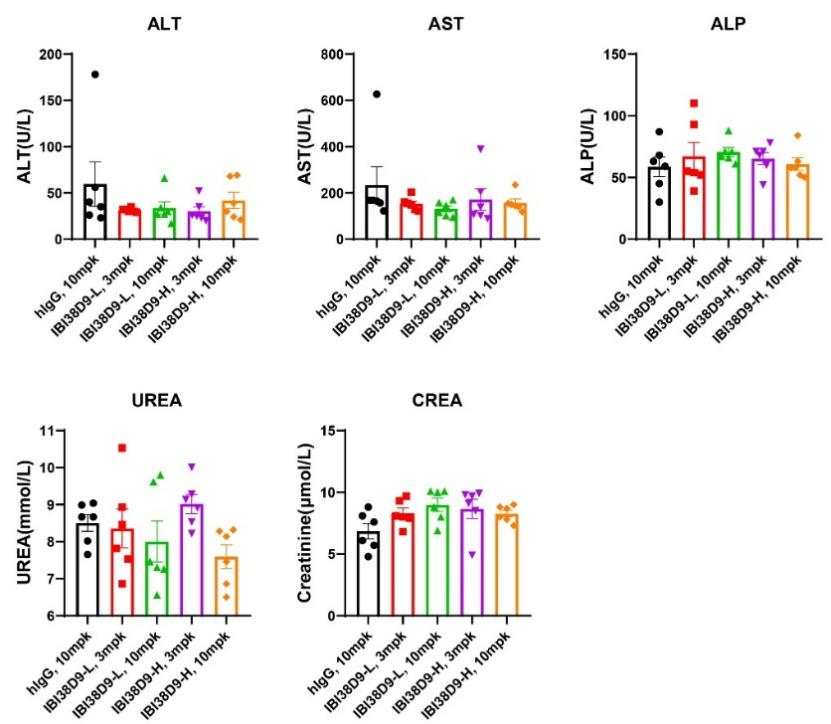
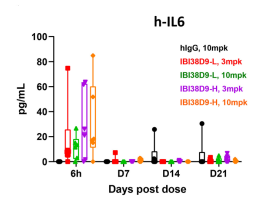
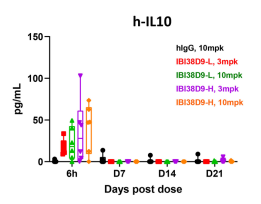
Fig 9. Preclinical toxicological evaluation of IBI38D9 dual-antagonist drug for HSC-NPG mice.
Mice were randomized into five groups (n=7 per group) based on peripheral T and B cell composition and treated with human IgG (10 mg/kg), IBI38D9-L (3 mg/kg and 10 mg/kg), or IBI38D8-H (3 mg/kg or 10 mg/kg) on days 0, 7, 14, and 21. Body weights were measured every other day and blood was collected 6 hours after administration for determination of cytokine and PD parameters. Mice were euthanized on day 35 and whole blood or serum was used for hemocyte or blood biochemical parameter analysis. The results showed that clearance of human B cells and depletion of T cells were observed in mice after administration of IBI38D9-L dual antibody, demonstrating the role of IBI38D9-L dual antibody in effectively killing CD79b-positive cells by coupling and activating T cells. Slight weight loss and mild factor release storm phenomenon were observed in the mouse model, indicating that the Hu-HSC-NPG model can reflect the toxicological effects of such dual-antibody drugs to a certain extent, and provide an important reference for drug clinical trials.
6. HSC-NPG infected with HIV applied in gene knockout therapy research[4]
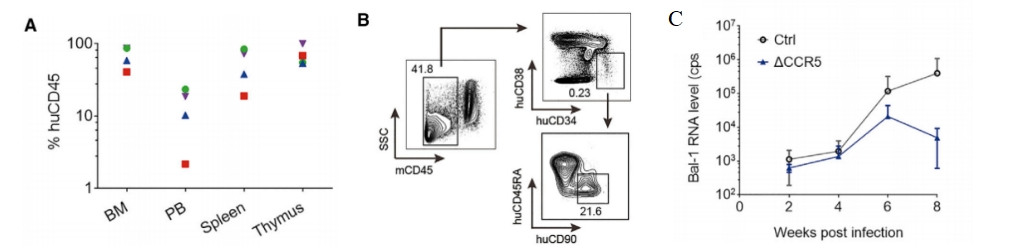
Fig10. Proportion of huCD45 in each organ after 12 weeks of reconstruction and Bal-1 RNA level after HIV infection in Hu-HSC-NPG secondary transplanted mice.
Newborn NPG mice were subjected to sublethal irradiation (1.0 Gy) and transplanted with 1×106 hCD34 HSC with a CCR5 gene knockout. After 12 weeks of reconstitution, bone marrow cells were collected for secondary transplantation. Four 6-8 week-old NPG mice underwent sublethal irradiation (1.6 Gy) and were transplanted with one-third of the bone marrow CD34 cells. After 12 weeks of reconstitution, the proportion of hCD45 in the bone marrow, peripheral blood, spleen, and thymus was detected. In the bone marrow, human CD34+/38-RA-90+ HSCs were detected. Further, HIV challenge experiments were conducted by intraperitoneal injection of HIV virus every two weeks to detect HIV Bal-1 RNA levels, and the mice were euthanized at week 8. The results showed that: secondary transplanted HSC-NPG mice had high levels of hCD45 cells in the bone marrow, peripheral blood, spleen, and thymus. At the same time, the CCR5 gene knockout HSC-NPG group had reduced HIV RNA levels, providing evidence for the use of CCR5 gene-edited HSC transplantation in clinical HIV therapy.
HuHSC-NPG Mice Applications
1. Human cell or tissue transplantation (e.g. human colon cancer tissue)
2. Tumor and tumor stem cell research (e.g. human lymphoma, human pancreatic cancer)
3. ES and iPS cell research
4. Hematopoietic and immunology research
5. Human disease infection model research (e.g. HIV, HIV, EB, CMV and other viral infection infections)
6.Research and development of humanized animal models
7. Evaluation of immunomodulatory drugs such as immune checkpoints (ICI)
8. Evaluation of tumor efficacy of antibody drugs targeting T cells, such as bispecific antibodies (BiTE).
9. Tumor immune microenvironment research
10.Cytokine release storm (CRS) modeling
Reference
1. Huang C, et al. Gastric cancer mesenchymal stem cells via the CXCR2/HK2/PD-L1 pathway mediate immunosuppression. Gastric Cancer. 2023, 26(5):691-707.
2. Liang S, et al. Identification of the immunosuppressive effect of γδ T cells correlated to bone morphogenetic protein 2 in acute myeloid leukemia. Front Immunol. 2022, 13:1009709.
3. Wang J, et al. Characterization of anti‑CD79b/CD3 bispecific antibody, a potential therapy for B cell malignancies. Cancer Immunol, Immunother 2023; 72:493-507.
4. Xu L, et al. CRISPR/Cas9-Mediated CCR5 Ablation in Human Hematopoietic Stem/Progenitor Cells Confers HIV-1 Resistance In Vivo. Mol Ther. 2017, 25(8):1782-1789.

 animalmodel@vital-bj.com
animalmodel@vital-bj.com +8610-84928167
+8610-84928167