General Information
Strain Name | B6-Tg HRAS9/Vst |
Common Name | RasH9; rasH |
Origin | Beijing Vitalstar Biotechnology Co., Ltd. |
Background | C57BL/6NCrl |
Coat Color | Black |
Edited gene | HRAS |
Development
Vitalstar constructed a chromosomal DNA fragment containing the coding sequence and introns of the human HRAS gene, upstream and downstream non-coding regions (the total length of the fragment is about 7.5 Kb, and the sequence of the last intron of the fragment was sentinel mutated into a sequence that enhances the gene expression, so that the gene can be expressed under the drive of the HRAS self-regulatory sequences), and injected into the prokaryotic nucleus of C57BL/6 mice. Positive founder mice were amplified to establish a line, from which a ninth line (named rasH9) was selected with the transgene integrated on mouse chromosome 6 and a transgene copy number of four. The expression was detected by western blot, and the results showed that HRAS was expressed in multiple organs of mice. It was demonstrated that the RasH9 transgenic mice could also be used in drug tumorigenicity assessment experiments.
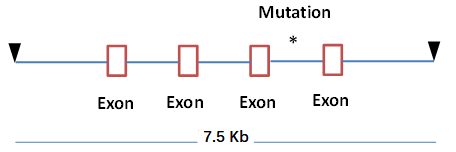
Fig 1. Schematic diagram of the RasH9 mouse construction strategy
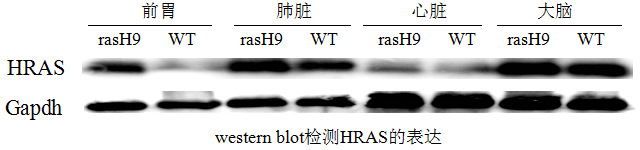
Fig 2. Western blot detection of RasH9 mice expression in various organs
Genotype identification information
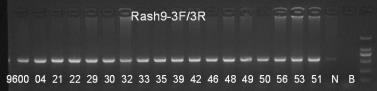
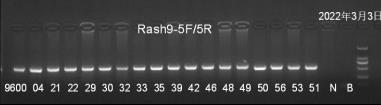
Fig3. RasH9 mouse genetic assay
Phenotype
Carcinogenicity experiments
Carcinogenicity experimental evaluation program:
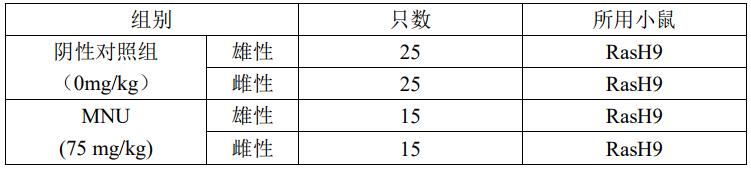
A single intraperitoneal injection of 75 mg/kg of MNU was given in the test group, and the day of administration was D1.
Growth was recorded throughout and all animals were systematically dissected and grossly observed at the end of the experiment. The examination at autopsy included body surface observation, all grossly diseased tissues with tumorigenesis.
1. Changes in body weight of each group

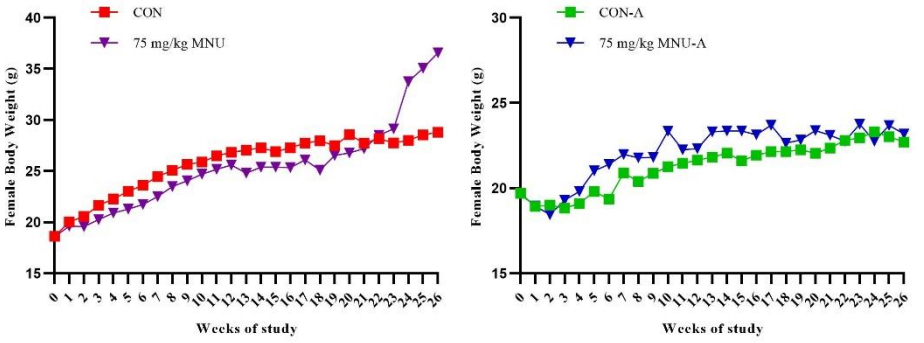
Fig 1. Changes in body weight of each group of mice in the carcinogenicity evaluation test
Note: In the figure, CON and 75 mg/kg MNU groups are RasH9 mice; CON-A and 75 mg/kg MNU-A groups are similar mice.
The results showed that the body weight of RasH9 male rats decreased after 126 days of MNU administration and increased significantly after 154 days of age in females compared to controls.
2. Survival rate of RasH9 mice in each group
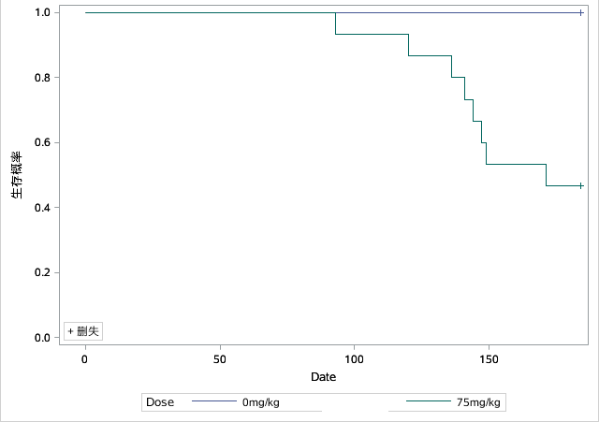
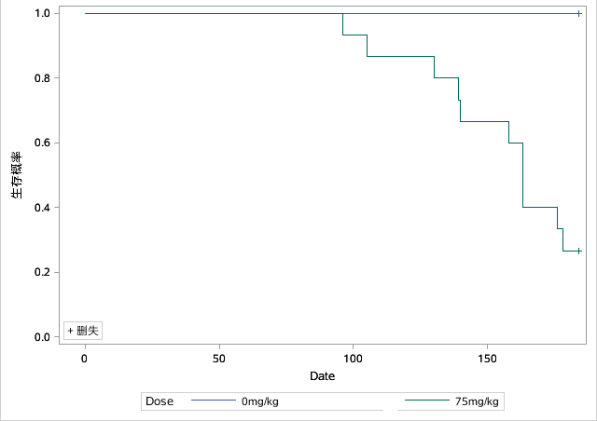
Fig 2. Survival curves in various groups of the carcinogenicity evaluation test (The left graph was male and the right graph was female)
Compared with controls, male RasH9 mice began to show mortality on day 93 and had a terminal survival rate of 46.7% at day 171; female RasH9 mice began to show mortality on day 96 and had a terminal survival rate of approximately 26.67% at day 178.
3. Tumors in RasH9 mice in each group of the carcinogenicity evaluation test
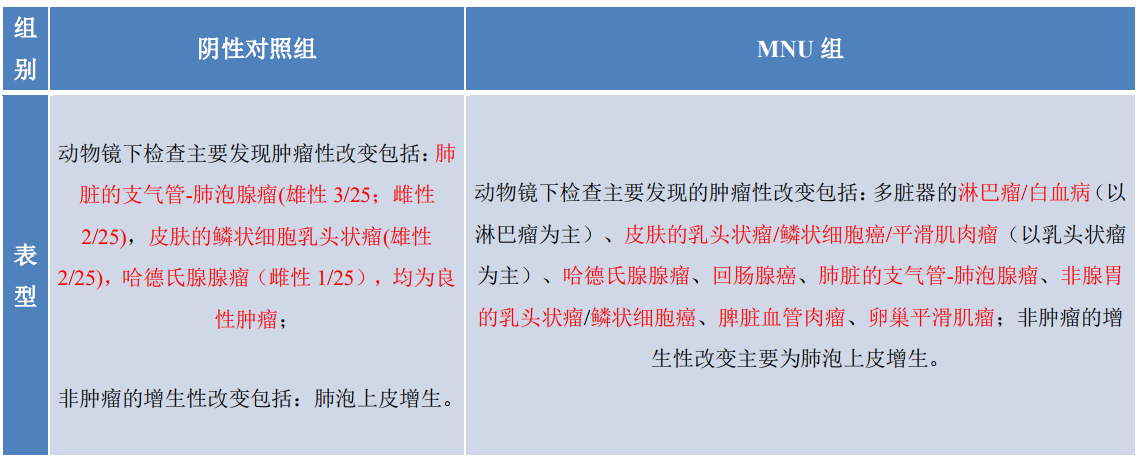
4. Tumors in MNU-treated RasH9 mice

NA = Not applicable or not reported in the literature;
*Includes lymphomas of the thymus, spleen, and lymph nodes originating in the hematopoietic system.
Under the conditions of this test, RasH9 mice given 75 mg/kg of MNU by a single intraperitoneal injection had essentially the same major tumor types and incidence as reported in the literature for similar mice as well as background values from our laboratory.
5. Histopathology of RasH9 mice in the carcinogenicity evaluation test group
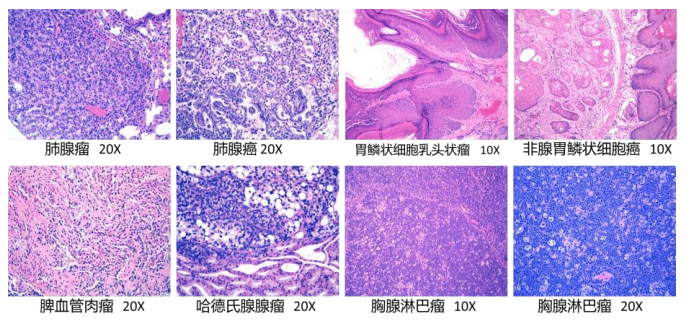
Figure 3. Tissue pathology of MUN group RasH9 mice in carcinogenicity assessment experiments.
Reference
1. Madhav G, et al. Historical control data of spontaneous tumors in transgenic CByB6F1-Tg(HRAS)2Jic(Tg.rasH2) mice. International Journal of Toxicology, 32: 48-57, 2013.
2. Nambiar P. R, et al. The rasH2 mouse model for assessing carcinogenic potential of pharmaceuticals. Toxicologic pathology, 41: 1058-1067, 2013.
3. Masaya T, et al. Interlaboratory comparison of short-term carcinogenicity studies using CB6F1-rasH2 transgenic mice. Toxicologic Pathology, 31: 191-199, 2003.
4. Prashant R, et al. Spontaneous tumour incidence in rasH2 mice: review of internal data and published literature. Toxicologic pathology, 40: 614-623, 2012.
5. Mansell K, et al. Evaluation of background data from rasH2 mice used in 26-week carcinogenicity. Charles River laboratiries preclinical services, 2013.

 animalmodel@vital-bj.com
animalmodel@vital-bj.com +8610-84928167
+8610-84928167